Perioperative analgesia in arthroscopic surgery of the knee
Analgesia perioperatoria en cirugía artroscópica de rodilla
Resumen:
Un adecuado control del dolor perioperatorio es clave no solo para brindar al paciente un máximo bienestar tras el procedimiento, sino por la implicación que tiene este en el grado de satisfacción que se obtiene al final del proceso quirúrgico.
Debido a la variedad de opciones analgésicas y ante la falta de consenso sobre la técnica ideal para el control del dolor tras realizar una artroscopia de rodilla, se plantea una revisión de la literatura. El objetivo es evaluar las diferentes herramientas que existen en la actualidad en función de la evidencia.
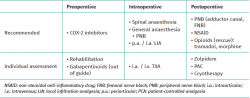
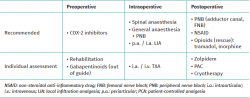
Para optimizar los resultados y lograr un precoz control de la extremidad, se sugiere al paciente trabajar su masa muscular cuadricipital, a la vez que se recomienda la utilización de antiinflamatorios no esteroideos (AINE) inhibidores de la COX-2 durante el preoperatorio.
Para el momento quirúrgico, junto con las diferentes alternativas anestésicas, se recomienda la realización de bloqueos nerviosos periféricos. Entre los bloqueos recomendados se encuentran el del canal del aductor y del nervio femoral. La realización de infiltraciones anestésicas locales ha demostrado su beneficio no solo por el acceso a los compartimentos laterales y anterior de la rodilla, sino por la posibilidad de acceder a la cápsula articular posterior. A su vez, la utilización del ácido tranexámico mediante infiltración intraarticular o intravenoso permite disminuir los episodios de hemartrosis y, por lo tanto, el dolor postoperatorio.
Con el objetivo de disminuir el requerimiento de opioides intravenosos en el postoperatorio, el uso de protocolos de analgesia multimodal ha ido ganando terreno. Es así que, junto a los bloqueos nerviosos periféricos, la administración de AINE, los sistemas de analgesia controlada por el paciente, la utilización de agentes como el zolpidem y la crioterapia forman parte del esquema recomendado para un correcto control del dolor tras una artroscopia de rodilla.
Abstract:
Adequate perioperative pain control is essential not only to ensure maximum patient well-being after the procedure, but also in view of the influence it has upon patient satisfaction at the end of the surgical process.
Due to the variety of analgesic options and the lack of consensus regarding which technique is best suited for pain control after knee arthroscopy, we decided to conduct a literature review on the subject. The aim was to assess the different tools available, based on the existing evidence.
In order to optimise the outcomes and secure early control of the extremity, it is advisable for the patient to work his or her quadriceps muscle mass, together with the administration of COX-2 inhibiting non-steroidal anti-inflammatory drugs (NSAIDs) during the preoperative period.
At the time of surgery, and in addition to the different anaesthetic alternatives, the use of peripheral nerve blocks is advised. The recommended block procedures include adductor canal block and femoral nerve block. Local anaesthetic infiltrations have demonstrated benefit not only in terms of access to the lateral and anterior compartments of the knee, but also in allowing access to the posterior joint capsule. In turn, the use of tranexamic acid as an intraarticular infiltration or via the intravenous route is able to reduce episodes of haemarthrosis and therefore of postoperative pain.
The use of multimodal analgesia protocols has gained acceptance with the purpose of reducing the need for intravenous opioids in the postoperative period. Accordingly, these protocols, together with peripheral nerve blocks, the administration of NSAIDs, patient-controlled analgesia systems, the use of drugs such as zolpidem, and cryotherapy, currently form part of the recommended strategy for correct pain control following knee arthroscopy.
Introduction
Uncontrolled pain is the most frequent complication following arthroscopic surgery of the knee. The absence of optimum pain management can increase postoperative morbidity, reduce patient satisfaction and have a negative impact upon the outcome of the procedure. Postoperative pain control is therefore extremely important for orthopaedic surgeons(1).
Poor postoperative pain management can delay patient discharge, resulting in a prolongation of hospital stay and an increased use of healthcare resources(2).
A number of techniques are currently available for the treatment of pain following arthroscopic surgery of the knee. The most widely used options include opioids, non-steroidal anti-inflammatory drugs (NSAIDs), and anaesthetics such as lidocaine or bupivacaine. The less commonly used strategies include ketamine, tranexamic acid (TXA), sedatives, gabapentinoids and corticosteroids(3).
Although historically the opiates and opioids were the gold standard for the control of postoperative pain, the complications of prolonged narcotic use have changed the paradigm of pain management in orthopaedic practice(4,5). The success of multimodal regimens has been well documented in the literature in relation to prosthetic surgery, with lower pain scores in the immediate postoperative period and even up to one year after the procedure(6,7).
The present narrative literature review summarises the most relevant information about the agents involved in perioperative pain management in the context of arthroscopic surgery of the knee, with the definition of clear evidence-based recommendations on the different available options.
Physiological principles of acute pain
Surgical procedures generate a great number of painful afferent signals, inducing a secondary inflammatory response that contributes considerably to postoperative pain. These signals are capable of initiating prolonged changes in both the central and the peripheral nervous system, leading to amplification and prolongation of postoperative pain(8). As a result of the inflammation at the surgical site, we can observe peripheral sensitisation, characterised by a decrease in the nociceptive threshold of the afferent terminals(9).
Pain assessment
The visual analogue scale (VAS), the numeric rating scale (NRS) and the verbal classification scale (VCS) are among the tools most widely used to assess pain intensity in clinical-surgical and research scenarios. Different studies have evaluated the usefulness of each of these instruments, with the identification of subtle differences between them. It is important to mention that, of the above scales, the NRS offers the greatest sensitivity, being able to detect differences between sexes and pain intensity(10).
Preoperative pain management
Defining objectives and establishing expectations
An important part of postoperative pain control begins long before surgery. A prospective study found that patients who were showed videos describing the treatment before the actual operation experienced significantly less pain 6 weeks later than those who did not see the videos(11). Preoperative assessment and the treatment of anxiety and depression are important, because it has been well established that these conditions are able to amplify pain perception(12).
Preventive analgesia
Preventive analgesia begins before the operation with the purpose of preventing sensitisation of the central and peripheral nervous system secondary to surgical incision and tissue manipulation. The prevention of such sensitisation can improve patient postoperative pain and reduce the risk of developing chronic neuropathic pain(8). Preventive analgesics should be relatively easy to administer, afford rapid action, and have adverse effects that do not interfere with the planned surgical procedure. Non-steroidal anti-inflammatory drugs, cyclooxygenase-2 (COX-2) inhibitors and paracetamol are generally used for this purpose, and are administered in the preoperative waiting room 1-2 hours before the start of surgery(13).
Cyclooxygenase-2 inhibitors
These drugs act at peripheral level, inhibiting the production of prostaglandins. They have a favourable adverse effects profile, with a reduced risk of gastric ulceration and minimal platelet alteration compared with the traditional NSAIDs, which are non-specific COX-1 and COX-2 inhibitors. Although COX-2 inhibitors have been implicated in an increased risk of adverse cardiovascular events, celecoxib doses of up to 400 mg a day have not been shown to increase this risk(14,15).
Paracetamol
This drug is believed to act via different centrally-mediated pathways, even as a cannabinoid receptor agonist, as a COX-2 isoenzyme inhibitor, and as an agonist of transient receptor potential cation channel subfamily V member 1 — a central anti-nociceptor(16).
Physical therapy
Preoperative muscle strength is an important factor that can influence the postoperative outcomes. One, two and three months after anterior cruciate ligament (ACL) reconstruction, those patients with preoperative strength > 90% on comparing the injured extremity versus the contralateral extremity, had significantly better strength than those patients with preoperative strength < 75%. Level of evidence 1 has shown that preoperative rehabilitation can facilitate postoperative recovery(17).
Intraoperative pain management
General and spinal anaesthesia
General anaesthesia is associated with a decrease in perioperative tissue oxygen tension(18), as well as with postoperative nausea, vomiting and delirium that can be avoided with the use of spinal anaesthesia(19). However, the administration of spinal anaesthesia requires technical skill, and although the procedure is generally very successful, the associated failure rate is 4%, requiring conversion to general anaesthesia(20). The complications rate of spinal anaesthesia is reported to be extremely low (0.03%). The most common adverse effects include postoperative hypotension and urinary retention(21).
Local infiltration anaesthesia (LIA)
Analgesia through local infiltration in the form of intraarticular (i.a.) or periarticular (p.a.) injections is another effective pain control strategy. Different studies have shown that local infiltration anaesthesia affords a significant decrease in pain and opioid consumption following ACL reconstruction surgery(22), with an effect not significantly different from that of femoral nerve block (FNB)(23,24,25). In addition, i.a. infiltration has been seen to have a lesser impact upon quadriceps muscle strength than FNB(26).
Different drug combinations can be used — the most common options being lidocaine, bupivacaine, ketorolac and morphine(22).
Periarticular and intraarticular LIA is effective in reducing postoperative pain after ACL reconstruction surgery, and may play a crucial role in multimodal pain control regimens. A randomised clinical trial (RCT) carried out by Koh et al. randomised 100 patients to 5 groups: 1) control group without injection; 2) i.a. ropivacaine; 3) i.a. multimodal drug cocktail (MDC); 4) p.a. MDC injection; and 5) i.a. + p.a. MDC injection. Five p.a. injections were distributed in the periosteum around the tendon extraction area, incision zone, patellar retinaculum, infrapatellar fat pad and in the periosteum and fascia around the tibial tunnel. The authors recorded significant pain reduction on the first postoperative night in the p.a. MDC group and i.a. + p.a. MDC group, compared with the other three cohorts (p < 0.001). This study suggested that p.a. injections may be more beneficial than i.a. injections for perioperative pain control(27).
Ischaemia cuff
Studies have been made of the role of the ischaemia cuff in the development of postoperative pain. The main variables in these studies are the duration of cuff use and the cuff pressure(28). Ischaemia produces tissue hypoxia and acidosis, proportional to its duration(29). Cuff times of over two hours can result in post-cuff syndrome with oedema, stiffness, paleness, weakness and subjective numbness that generally resolve within one week. Minor complications can also be observed, such as weakness of the quadriceps, which is probably of a multifactorial nature(30).
Tranexamic acid
Tranexamic acid has been found to be promising in reducing pain intensity and the incidence of haemarthrosis in the early postoperative period following arthroscopy of the knee. This molecule is a synthetic antifibrinolytic derivative of lysine; it competitively inhibits fibrinolysis and stabilises clot formation(31). Although there are different intravenous (i.v.) TXA dosing regimens, recent studies have found the desired haemostatic and analgesic effects to be achieved using doses of 10 and 15 mg/kg(32,33). It has been demonstrated that both i.a. and i.v. administration significantly reduce pain and the joint effusion volume of the knee, without increasing the risk of thromboembolic events. Furthermore, i.a. and i.v. administration are comparable in terms of efficacy, yielding similar VAS scores in the first and second postoperative weeks(34).
Postoperative pain management
Multimodal analgesia
With the decrease in use of opioids associated to the shift in pain treatment paradigm towards a multimodal approach, a range of analgesic drug combinations and regimens have been developed and tested.
Multimodal analgesia refers to the combination of different types of drugs and administration routes, including peripheral nerve block (PNB), p.a. injection, patient-controlled analgesia (PCA), and drugs administered via the oral route — both opioids and non-opioids. The aim of multimodal analgesia is to afford superior postoperative pain control through the simultaneous modulation of various pain pathways, with minimisation of the undesired adverse effects of excessive opioid use(35).
A recent multicentre study compared different approaches to the management of pain after ACL reconstruction in terms of subjective pain as assessed by the VAS, opioid consumption, late discharge rates and complications. The analgesic procedures included in the study were continuous and single dose FNB, and analgesia using the p.a. or i.a. LIA technique. Comparison was also made of the use of NSAIDs and corticosteroids via the intravenous route. The patients who received NSAIDs were less likely to show delays in discharge, and the use of dexamethasone was associated to a decrease in opioid consumption and fewer side effects of the nerve block agents(35).
Opiates and opioids
Opiates and their synthetic analogues, the opioids, were among the most widely used options for pain control in the acute postoperative phase of a large range of orthopaedic procedures.
As analgesics, the opioids inhibit pain signal conduction through the activation of their receptors via different methods of administration.
Although in the past the opioids were more often used as the principal pain control measure following arthroscopic surgery of the knee (e.g. reconstruction of the ACL), their potential for abuse or addiction, the withdrawal symptoms following prolonged use, and other unpleasant side effects such as diminished intestinal motility, have caused pain management strategies to shift towards a more multimodal approach in recent years(36).
Nevertheless, opiates are still often used to treat breakthrough pain within the first week after ACL reconstruction surgery, particularly when the pain cannot be controlled by alternative means. The most frequently prescribed opioids include tramadol and — less commonly — morphine(37).
A number of studies have explored the risk factors implicated in the prolonged use of opioids following ACL surgery, including a meta-analysis of 9,474 United States military personnel that received opioids following reconstruction of the ACL. The analysis revealed that 28% of the study population continued to take opioids for over 90 days, and found the number and frequency of pre- and perioperative prescriptions to be the main risk factor in the prolonged postoperative use of these drugs(38).
Given the risk of patient drug dependence, the paradigm of pain management has shifted in recent years towards more multimodal treatment regimens.
Peripheral nerve blocks
Arthroscopic surgery of the knee is most often performed in the ambulatory setting. In this regard, adequate pain control allowing opportune patient discharge and satisfaction is essential for optimising care and reducing needless costs associated with delays in discharge or patient readmission(39). Peripheral nerve block is increasingly used for perioperative pain control. The benefits of PNB include a decrease in intra- and postoperative opioid use, better postoperative pain control, and less fatigue and nausea associated with other oral forms of pain management. Side effects of PNB are rare, but include a small risk of nerve damage and prolonged muscle weakness(40).
The most widely used block procedures are femoral nerve block, adductor canal block and sciatic nerve block(41).
Femoral nerve block
Excluding the posterior capsule, the knee joint is innervated by the femoral nerve; this causes many surgeons to resort to femoral nerve block (FNB) for postoperative analgesia(42).
This block is targeted to the femoral nerve in the femoral canal, and depends on diffusion of the local anaesthetic to block the femoral nerve, lateral femoral cutaneous nerve and obturator nerve. Traditionally, the technique has been performed using nerve stimulation, though ultrasound is now increasingly employed to reduce the risk of failed block and of accidental arterial or intraneural puncture. Femoral nerve block is a mixed (motor and sensory) block procedure; it therefore causes numbness of the anterior, lateral and medial surfaces of the thigh, resulting in profound weakness of the quadriceps muscle. When this block is performed, the patient is placed in supine decubitus, and ultrasound is used to identify the nerve at the confluence of the iliac and psoas muscles. Ultrasound is also used to visualise the local anaesthetic injected below and around the femoral nerve(43).
In a RCT of patients subjected to ACL reconstruction under spinal anaesthesia with or without FNB, the patients subjected to block reported lesser pain — though the difference only proved significant after 6 hours (p = 0.007). However, following hospital discharge, the patients who had undergone FNB were seen to suffer increased pain as the effect of the block subsided(44).
The literature offers no proof of the superiority of FNB over other analgesia modes. In a systematic review of the use of FNB in ACL reconstruction surgery, Mall and Wright recorded no evidence of additional benefit compared with multimodal analgesia(45).
A number of studies have suggested that FNB can affect knee strength and function for a prolonged period beyond the immediate postoperative stage(46,47,48). The estimated prevalence of transient or even permanent neurological deficits is 1.94%(49).
Adductor canal block
This block technique is fundamentally targeted to the sensory nerves in the adductor canal, with a reduction in the degree of weakness of the quadriceps muscle. It includes the saphenous nerve, the articular branches of the obturator nerve, the medial retinacular nerve and the nerve of the vastus medialis, which is the only motor nerve involved.
This technique results in sensory block of the anteromedial surface of the knee at the level of the upper pole of the patella and medial portion of the leg — with minimal loss of quadriceps muscle strength. This block is performed with the patient in supine decubitus, applying the ultrasound device on the medial surface of the thigh at middle-distal level and 2-3 cm proximal to the adductor hiatus. The femoral artery and vein are located in depth to the sartorius muscle, with the saphenous nerve running lateral to them at this level, and the local anaesthetic is injected around the nerve(50).
Abdallah et al., in a RCT, found adductor canal block to offer better analgesia than FNB in patients subjected to reconstruction of the ACL, since it ensures effective postoperative pain control and also causes lesser weakness of the quadriceps muscle(51).
Continuous infusion in nerve blocks
Catheters can be used to provide continuous FNB or perform i.a. injections or wound infiltrations. The use of such catheters has been shown to afford effective pain relief after ACL reconstruction surgery, as well as improved early physical rehabilitation(52,53).
In contrast to the findings of the above-mentioned studies, different authors have concluded that this technique offers no clinical advantages and is associated to a considerable incidence of complications (up to 13%)(54).
Non-steroidal anti-inflammatory drugs and selective COX-2 inhibitors
Non-steroidal anti-inflammatory drugs are commonly used as analgesics in orthopaedic surgery and also regularly in postoperative multimodal pain management protocols for surgery of the ACL(55).
Many studies have evaluated their efficacy as postoperative analgesia, and have shown NSAIDs to be both effective and safe(56,57).
In a triple-blind RCT, Mardani-Kivi et al. found the use of celecoxib as preventive analgesia following ACL reconstruction to reduce pain intensity and opioid consumption in the celecoxib group 6 and 24 hours after surgery (p < 0.0001)(58). In addition, NSAIDs are associated to fewer side effects such as nausea and vomiting, and are better tolerated by patients than other postoperative analgesics(59,60).
Previous studies have suggested that NSAIDs may have adverse effects in terms of patient recovery and healing after orthopaedic procedures(61,62). However, other studies have reported no negative effects with the use of NSAIDs and selective COX-2 inhibitors in terms of soft tissue and tendon healing(63). The studies that have evaluated the use of NSAIDs in repair of the ACL, shoulder/labrum and meniscus likewise have recorded no differences in healing rate in relation to NSAID use during the postoperative phase(63,64).
Ketamine
Ketamine is a phencyclidine derivative with antagonistic action upon the N-methyl-D-aspartate (NMDA) receptors(65).
In a randomised prospective study, Zhu et al. evaluated the analgesic effect of mixing ketamine with ropivacaine in combined sciatic and femoral nerve blocks guided by ultrasound (SFNB)(66). The pain scores were significantly lower in the RNK group (ropivacaine + perineural ketamine) versus the RIK group (ropivacaine + i.v. ketamine) and the R group (ropivacaine alone) at 20 and 24 hours post-surgery (p = 0.001). In addition, the RNK group took longer in requiring the first analgesic dose (p = 0.014) compared with the other two groups — suggesting that perineural ketamine improves the antinociceptive effect of SFNB(66).
Sedatives and corticosteroids
Although less frequently used in clinical practice, sedatives, gabapentin and corticosteroids have been investigated as alternatives for the management of pain in arthroscopy of the knee. The sedatives described in postoperative pain treatment following reconstruction of the ACL are typically of the non-benzodiazepine class, including agents such as zolpidem(67).
It has been reported that patients administered 10 mg of zolpidem each night during the first postoperative week show a 28% decrease in opioid use(68).
Gabapentin and pregabalin have been used as anticonvulsants and analgesics, particularly in cases where the pain is believed to be of neuropathic origin(69,70).
Gabapentin administered preoperatively has been investigated as a potential option in multimodal pain management. Randomised clinical trials have been carried out involving 150 mg of pregabalin administered pre- and postoperatively, with the observation of a decrease in subjective pain intensity as measured by the VAS, and in postoperative opioid use(71). However, these results come into conflict with the findings of another RCT on the pre- and postoperative use of pregabalin in ACL reconstruction that found the drug to be no better than placebo in reducing postoperative pain and opioid use(72).
Given the risk of immune suppression and other side effects, the role of corticosteroids in pain control is relatively limited in arthroscopic surgery of the knee. It has been shown that the use of NSAIDs together with 8 mg of i.v. dexamethasone in the postoperative period of ACL reconstruction surgery is superior to NSAIDs alone in reducing patient pain(73). In addition, adding 1-4 mg of dexamethasone to the bupivacaine solution when performing subsartorial saphenous nerve block (SSNB) potentially increases the duration of block by up to 13 hours(73).
Patient-controlled analgesia
Patient-controlled analgesia is an option for the management of postoperative pain in knee arthroscopy, since it affords simple, rapid and adequate pain relief. It can be administered in different ways and requires a high level of collaboration on the part of the patient. The device is programmed depending on the analgesic used, and the physical characteristics and basal pain of the patients. The patient is able to administer a small amount of analgesic by pressing the button when needed. In general, the opioids used in PCA are morphine, fentanyl and tramadol(74).
Although the technique has some adverse effects such as nausea, vomiting, respiratory depression and urinary retention, they are less severe than those associated with conventional opioid treatment. Consequently, PCA is considered to be safe and effective in treating moderate to intense postoperative pain(75).
Cryotherapy
The effects of postoperative cryotherapy have been evaluated by a number of RCTs, of which approximately one-half documented a decrease in pain symptoms(76). Preoperative cryotherapy(77), as well as combined cryotherapy and compression(78), have also shown beneficial effects. It is believed that the therapeutic effect of cold is due to a decrease in soft tissue swelling and muscle spasms, as well as to action upon the pain threshold(79). Paralysis of the peroneal nerve has been reported as a complication of the prolonged application of ice to the knee. Such problems were transient and resolved spontaneously, however(80).
Continuous passive motion devices
The use of continuous passive motion (CPM) devices for postoperative pain relief is subject to controversy. Some studies have reported a decrease in narcotic use in patients subjected to CPM(81,82), while another has documented no effect upon analgesic use(83). A number of studies have shown that CPM does not affect range of motion or anterior laxity following reconstruction of the ACL(84). Given the cost related to CPM and its lack of impact upon the general functional outcomes, it is difficult to justify the use of this technique as a simple complement to pain control in the absence of solid support in the form of level 1 evidence(85).
Transcutaneous electrical nerve stimulation (TENS)
Transcutaneous electrical nerve stimulation is another complementary technique for pain control. In the central nervous system, TENS activates areas in the brainstem and spinal cord that contain serotonin, muscarinic and opioid receptors(86).
Following knee arthroscopy, patients subjected to TENS for an average of four days showed a decrease in postoperative pain that proved significant on the second day, with the recovery of preoperative mobility, isokinetic strength in extension and flexion, and leg volume one month earlier than the patients in the placebo group(87).
Table 1 summarises the aspects addressed in this narrative review on perioperative pain control in arthroscopic surgery of the knee.
Conclusions
The optimum management of postoperative pain following arthroscopic surgery of the knee remains subject to controversy, with no widely accepted approach having been established to date.
Based on the present review, the following recommendations can be made:
- Preoperative rehabilitation can facilitate postoperative recovery.
- The preventive administration of COX-2 inhibitors and gabapentinoids in the preoperative period is effective in reducing pain after knee arthroscopy.
- Periarticular and intraarticular LIA is effective in reducing postoperative pain and may play a crucial role in multimodal pain control regimens.
- The new approach focuses on multimodal analgesia with the aim of improving pain control in the immediate postoperative period using combinations of different drugs at low doses.
- The change in paradigm referred to pain control seeks to avoid increased use and potential abuse of opioids and opiates.
- Peripheral nerve blocks are the most effective analgesic measure, and thus should be regarded as the reference technique. It is advisable to choose an adductor canal block to avoid the quadriceps muscle weakness that would result from femoral block.
- Tranexamic acid has been found to be promising in reducing pain intensity and the incidence of haemarthrosis in the early postoperative period following arthroscopy of the knee.
As in all orthopaedic surgeries, the choice of analgesic protocol should be established on an individualised basis, seeking maximum effectiveness with the lowest possible incidence of adverse effects and complications.
Información del artículo
Cita bibliográfica
Autores
Maximiliano Ibañez
Hospital Universitario Quirón Dexeus. Barcelona
Laura Valls
Hospital Universitario de Igualada. Barcelona
Sergi Gil
Hospital Universitario de Igualada. Barcelona
Juan Ignacio Erquicia
Hospital Universitario de Igualada. Barcelona
iMove Traumatología. Clínica Mi Tres Torres. Barcelona
Ethical responsibilities
Conflicts of interest. The authors state that they have no conflicts of interest.
Financial support. This study has received no financial support.
Protection of people and animals. The authors declare that this research has not involved human or animal experimentation.
Data confidentiality. The authors declare that the protocols of their work centre referred to the publication of patient information have been followed.
Right to privacy and informed consent. The authors declare that no patient data appear in this article.
Referencias bibliográficas
-
1Chmielewski TL, Jones D, Day T, Tillman SM, Lentz TA, George SZ. The association of pain and fear of movement/reinjury with function during anterior cruciate ligament reconstruction rehabilitation. J Orthop Sports Phys Ther. 2008;38(12):746-53.
-
2United States Acute Pain Management Guideline Panel: Acute Pain Management: Operative or Medical Procedures and Trauma. Pub. no. 92-0032. Rockville, Maryland: United States Department of Health and Human Services, Public Health Service Agency for Health Care Policy and Research; 1992.
-
3Davey MS, Hurley ET, Anil U, et al. Pain Management strategies after anterior cruciate ligament reconstruction: a systematic review with network meta-analysis. Arthroscopy. 2021;37(4):1290-300.
-
4Malige A, Bram JT, Maguire KJ, McNeely LW, Ganley TJ, Williams BA. Decreased prescribing of postoperative opioids in pediatric ACL reconstruction: treatment trends at a single center. Orthop J Sports Med. 2021;9(2):2325967120979993.
-
5Morris BJ, Mir HR. The opioid epidemic: impact on orthopaedic surgery. JAAOS. 2015;23(5):267-71.
-
6Moucha CS, Weiser MC, Levin EJ. Current strategies in anesthesia and analgesia for total knee arthroplasty. J Am Acad Orthop Surg. 2016;24:60-73.
-
7Gaffney CJ, Pelt CE, Gililland JM, Peters CL. Perioperative pain management in hip and knee arthroplasty. Orthop Clin North Am. 2017;48:407-19.
-
8Woolf CJ, Chong MS. Preemptive analgesia — treating postoperative pain by preventing the establishment of central sensitization. Anesth Analg. 1993;77:362-79.
-
9Raja SN, Meyer RA, Campbell JN. Peripheral mechanisms of somatic pain. Anesthesiology. 1988;68:571-90.
-
10Ferreira Valente MA, Pais Ribeiro JL, Jensen MP. Validity of four pain intensity rating scales. Pain. 2011;152(10):2399-404.
-
11Maddison R, Prapavessis H, Clatworthy M. Modeling and rehabilitation following anterior cruciate ligament reconstruction. Ann Behav Med. 2006;31(1):89-98.
-
12Von Korff M, Simon G. The relationship between pain and depression. Br J Psychiatry Suppl. 1996;30:101-8.
-
13Jianda X, Yuxing Q, Yi G, Hong Z, Libo P, Jianning Z. Impact of preemptive analgesia on inflammatory responses and rehabilitation after primary total knee arthroplasty: a controlled clinical study. Sci Rep. 2016;6:30354.
-
14Graham DJ. COX-2 inhibitors, other NSAIDs, and cardiovascular risk: the seduction of common sense. JAMA. 2006;296(13):1653-6.
-
15Bhala N, Emberson J, Merhi A, et al. Vascular and upper gastrointestinal effects of non-steroidal anti-inflammatory drugs: meta-analyses of individual participant data from randomized trials. Lancet. 2013;382(9894):769-79.
-
16Mallet C, Barrière DA, Ermund A, et al. TRPV1 in brain is involved in acetaminophen-induced antinociception. PLoS One. 2010;5(9):e12748.
-
17Shelbourne KD, Wilckens JH, Mollabashy A, DeCarlo M. Arthrofibrosis in acute anterior cruciate ligament reconstruction. The effect of timing of reconstruction and rehabilitation. Am J Sports Med. 1991;19(4):332-6.
-
18Treschan TA, Taguchi A, Ali SZ, et al. The effects of epidural and general anesthesia on tissue oxygenation. Anesth Analg. 2003;96(6):1553-7.
-
19Pugely AJ, Martin CT, Gao Y, Mendoza-Lattes S, Callaghan JJ. Differences in shortterm complications between spinal and general anesthesia for primary total knee arthroplasty. J Bone Joint Surg Am. 2013;95(3):193-9.
-
20Fettes PD, Jansson JR, Wildsmith JA. Failed spinal anaesthesia: mechanisms, management, and prevention. Br J Anaesth. 2009;102(6):739-48.
-
21Horlocker TT. Complications of regional anesthesia and acute pain management. Anesthesiol Clin. 2011;29(2):257-78.
-
22Tetzlaff JE, Dilger JA, Abate J, Parker RD. Preoperative intra-articular morphine and bupivacaine for pain control after outpatient arthroscopic anterior cruciate ligament reconstruction. Reg Anesth Pain Med. 1999 May-Jun;24(3):220-4.
-
23Kristensen PK, Pfeiffer-Jensen M, Storm JO, Thillemann TM. Local infiltration analgesia is comparable to femoral nerve block after anterior cruciate ligament reconstruction with hamstring tendon graft: a randomized controlled trial. Knee Surg Sports Traumatol Arthrosc. 2014 Feb;22(2):317-23.
-
24Mayr HO, Entholzner E, Hube R, Hein W, Weig TG. Pre- versus postoperative intraarticular application of local anesthetics and opioids versus femoral nerve block in anterior cruciate ligament repair. Arch Orthop Trauma Surg. 2007 May;127(4):241-4.
-
25Mehdi SA, Dalton DJ, Sivarajan V, Leach WJ. BTB ACL reconstruction: femoral nerve block has no advantage over intraarticular local anaesthetic infiltration. Knee Surg Sports Traumatol Arthrosc. 2004 May;12(3):180-3.
-
26Iamaroon A, Tamrongchote S, Sirivanasandha B, et al. Femoral nerve block versus intra-articular infiltration: a preliminary study of analgesic effects and quadriceps strength in patients undergoing arthroscopic anterior cruciate ligament reconstruction. J Med Assoc Thai. 2016 May;99(5):578-83.
-
27Koh IJ, Chang CB, Seo ES, Kim SJ, Seong SC, Kim TK. Pain management by periarticular multimodal drug injection after anterior cruciate ligament reconstruction: a randomized, controlled study. Arthroscopy. 2012;28(5):649-57.
-
28Wakai A, Winter DC, Street JT, Redmond PH. Pneumatic tourniquets in extremity surgery. J Am Acad Orthop Surg. 2001;9(5):345-35.
-
29Flatt AE. Tourniquet time in hand surgery. Arch Surg. 1972;104(2):190-2.
-
30Saunders KC, Louis DL, Weingarden SI, Waylonis GW. Effect of tourniquet time on postoperative quadriceps function. Clin Orthop Relat Res. 1979;(143):194-9.
-
31Lin ZX, Woolf SK. Safety, efficacy, and cost-effectiveness of tranexamic acid in orthopedic surgery. Orthopedics. 2016;39(2):119-30.
-
32Chiang ER, Chen KH, Wang ST, et al. Intra-articular injection of tranexamic acid reduced postoperative hemarthrosis in arthroscopic anterior cruciate ligament reconstruction: a prospective randomized study. Arthroscopy. 2019;35(7):2127-32.
-
33Felli L, Revello S, Burastero G, et al. Single intravenous administration of tranexamic acid in anterior cruciate ligament reconstruction to reduce postoperative hemarthrosis and increase functional outcomes in the early phase of postoperative rehabilitation: a randomized controlled trial. Arthroscopy. 2019;35(1):149-57.
-
34Ma R, Wu M, Li Y, et al. The comparative efficacies of intravenous administration and intra-articular injection of tranexamic acid during anterior cruciate ligament reconstruction for reducing postoperative hemarthrosis: a prospective randomized study. BMC Musculoskelet Disord. 2021;22(1):114.
-
35Baverel L, Cucurulo T, Lutz C, et al. Anesthesia and analgesia methods for outpatient anterior cruciate ligament reconstruction. Orthop Traumatol Surg Res. 2016;102(8s):S251-s255.
-
36Soffin EM, Waldman SA, Stack RJ, Liguori GA. An evidence-based approach to the prescription opioid epidemic in orthopedic surgery. Anesth Analg. 2017;125(5):1704-13.
-
37Beck JJ, Cline K, Sangiorgio S, Serpa R, Shifflett KA, Bowen RE. Prospective Study of acute opioid use after adolescent anterior cruciate ligament reconstruction shows no effect from patient- or surgical-related factors. J Am Acad Orthop Surg. 2020;28(7):293-300.
-
38Anderson AB, Balazs GC, Brooks DI, Potter BK, Forsberg JA, Dickens JF. Prescription patterns and risk factors for prolonged opioid dependence in elective anterior cruciate ligament reconstruction in a military population. Orthop J Sports Med. 2020;8(6):2325967120926489.
-
39Jansson H, Narvy SJ, Mehran N. Perioperative pain management strategies for anterior cruciate ligament reconstruction. JBJS Rev. 2018;6(3):e3.
-
40Vorobeichik L, Brull R, Joshi GP, Abdallah FW. Evidence basis for regional anesthesia in ambulatory anterior cruciate ligament reconstruction: part I-femoral nerve block. Anesth Analg. 2019;128(1):58-65.
-
41Min H, Ouyang Y, Chen G. Anterior cruciate ligament reconstruction with the use of adductor canal block can achieve similar pain control as femoral nerve block. Knee Surg Sports Traumatol Arthrosc. 2020;28(8):2675-86.
-
42Williams BA, Kentor ML, Vogt MT, et al. Reduction of verbal pain scores after anterior cruciate ligament reconstruction with 2-day continuous femoral nerve block: a randomized clinical trial. Anesthesiology. 2006;104(2):315-27.
-
43American Society of Anesthesiologists Task Force on Acute Pain Management: Practice guidelines for acute pain management in the perioperative setting: An updated report by the American Society of Anesthesiologists Task Force on Acute Pain Management. Anesthesiology. 2012;116(2):248-73.
-
44Astur DC, Aleluia V, Veronese C, et al. A prospective double blinded randomized study of anterior cruciate ligament reconstruction with hamstrings tendon and spinal anesthesia with or without femoral nerve block. Knee. 2014 Oct;21(5):911-5.
-
45Mall NA, Wright RW. Femoral nerve block use in anterior cruciate ligament reconstruction surgery. Arthroscopy. 2010 Mar;26(3):404-16.
-
46Magnussen RA, Pottkotter K, Stasi SD, et al. Femoral nerve block after anterior cruciate ligament reconstruction. J Knee Surg. 2017 May;30(4):323-8.
-
47Luo TD, Ashraf A, Dahm DL, Stuart MJ, McIntosh AL. Femoral nerve block is associated with persistent strength deficits at 6 months after anterior cruciate ligament reconstruction in pediatric and adolescent patients. Am J Sports Med. 2015 Feb;43(2):331-6.
-
48Swank KR, DiBartola AC, Everhart JS, Kaeding CC, Magnussen RA, Flanigan DC. The effect of femoral nerve block on quadriceps strength in anterior cruciate ligament reconstruction: a systematic review. Arthroscopy. 2017 May;33(5):1082-91.e1.
-
49Widmer B, Lustig S, Scholes CJ, et al. Incidence and severity of complications due to femoral nerve blocks performed for knee surgery. Knee. 2013 Jun;20(3):181-5.
-
50Manickam B, Perlas A, Duggan E, Brull R, Chan VW, Ramlogan R. Feasibility and efficacy of ultrasound-guided block of the saphenous nerve in the adductor canal. Reg Anesth Pain Med. 2009;34(6):578-80.
-
51Abdallah FW, Whelan DB, Chan VW, et al. Adductor canal block provides noninferior analgesia and superior quadriceps strength compared with femoral nerve block in anterior cruciate ligament reconstruction. Anesthesiology. 2016 May;124(5):1053-64.
-
52Dauri M, Fabbi E, Mariani P, et al. Continuous femoral nerve block provides superior analgesia compared with continuous intra-articular and wound infusion after anterior cruciate ligament reconstruction. Reg Anesth Pain Med. 2009 Mar-Apr;34(2):95-9.
-
53Vintar N, Rawal N, Pohar M, Veselko M. Intraarticular patient-controlled analgesia improves early rehabilitation after knee surgery. Coll Antropol. 2010 Sep;34(3):941-5.
-
54Woods GW, O’Connor DP, Calder CT. Continuous femoral nerve block versus intraarticular injection for pain control after anterior cruciate ligament reconstruction. Am J Sports Med. 2006 Aug;34(8):1328-33.
-
55Moutzouros V, Jildeh TR, Khalil LS, et al. A Multimodal protocol to diminish pain following common orthopedic sports procedures: can we eliminate postoperative opioids? Arthroscopy. 2020;36(8):2249-57.
-
56Bourne MH. Analgesics for orthopedic postoperative pain. Am J Orthop (Belle Mead NJ). 2004;33(3):128-35.
-
57Beck PR, Nho SJ, Balin J, et al. Postoperative pain management after anterior cruciate ligament reconstruction. J Knee Surg. 2004;17(1):18-23.
-
58Mardani-Kivi M, Karimi Mobarakeh M, Haghighi M, et al. Celecoxib as a preemptive analgesia after arthroscopic knee surgery; a triple-blinded randomized controlled trial. Arch Orthop Trauma Surg. 2013;133(11):1561-6.
-
59Secrist ES, Freedman KB, Ciccotti MG, Mazur DW, Hammoud S. Pain Management after outpatient anterior cruciate ligament reconstruction: a systematic review of randomized controlled trials. Am J Sports Med. 2016;44(9):2435-47.
-
60Ge H, Liu C, Shrestha A, Wu P, Cheng B. Do nonsteroidal anti-inflammatory drugs affect tissue healing after arthroscopic anterior cruciate ligament reconstruction? Med Sci Monitor. 2018;24:6038-43.
-
61Tsai WC, Hsu CC, Chen CP, Chen MJ, Lin MS, Pang JH. Ibuprofen inhibition of tendon cell migration and down-regulation of paxillin expression. J Orthop Res. 2006;24(3):551-8.
-
62Li Q, Zhang Z, Cai Z. High-dose ketorolac affects adult spinal fusion: a meta-analysis of the effect of perioperative nonsteroidal anti-inflammatory drugs on spinal fusion. Spine. 2011;36(7):E461-8.
-
63Blomquist J, Solheim E, Liavaag S, Baste V, Havelin LI. Do nonsteroidal anti-inflammatory drugs affect the outcome of arthroscopic Bankart repair? Scand J Med Sci Sports. 2014;24(6):e510-4.
-
64Proffen BL, Nielson JH, Zurakowski D, Micheli LJ, Curtis C, Murray MM. The Effect of perioperative ketorolac on the clinical failure rate of meniscal repair. Orthop J Sports Med. 2014;2:5.
-
65Peltoniemi MA, Hagelberg NM, Olkkola KT, Saari TI. Ketamine: a Review of clinical pharmacokinetics and pharmacodynamics in anesthesia and pain therapy. Clin Pharmacokinet. 2016;55(9):1059-77.
-
66Zhu T, Gao Y, Xu X, Fu S, Lin W, Sun J. Effect of ketamine added to ropivacaine in nerve block for postoperative pain management in patients undergoing anterior cruciate ligament reconstruction: a randomized trial. Clin Ther. 2020;42(5):882-91.
-
67Tompkins M, Plante M, Monchik K, Fleming B, Fadale P. The use of a non benzodiazepine hypnotic sleep-aid (Zolpidem) in patients undergoing ACL reconstruction: a randomized controlled clinical trial. Knee Surg Sports Traumatol Arthrosc. 2011;19(5):787-91.
-
68O’Hagan ET, Hübscher M, Miller CB, et al. Zolpidem reduces pain intensity postoperatively: a systematic review and meta-analysis of the effect of hypnotic medicines on post-operative pain intensity. Syst Rev. 2020;9(1):206.
-
69Wiffen PJ, Derry S, Bell RF, et al. Gabapentin for chronic neuropathic pain in adults. Cochrane Database Syst Rev. 2017;6(6):CD007938.
-
70Sills GJ. The mechanisms of action of gabapentin and pregabalin. Curr Opin Pharmacol. 2006;6(1):108-13.
-
71Kavak Akelma F, Baran Akkuş I, Altinsoy S, Özkan D, Ergil J. The effects of pregabalin and adductor canal block on postoperative pain in arthroscopic anterior cruciate ligament reconstruction. Turk J Med Sci. 2020;50(1):195-204.
-
72Nimmaanrat S, Tangtrakulwanish B, Klabklay P, Boonriong T. Perioperative administration of pregabalin in patients undergoing arthroscopic anterior cruciate ligament reconstruction: does it help to relieve postoperative pain? J Med Assoc Thai. 2012;95(10):1297-301.
-
73Chisholm MF, Cheng J, Fields KG, et al. Perineural dexamethasone with subsartorial saphenous nerve blocks in ACL reconstruction. Knee Surg Sports Traumatol Arthrosc. 2017;25(4):1298-306.
-
74Walder B, Schafer M, Henzi I, Tramèr MR. Efficacy and safety of patient controlled opioid analgesia for acute postoperative pain. A quantitative systematic review. Acta Anaesthesiol Scand. 2001;45:795-804.
-
75Dias AS, Rinaldi T, Barbosa LG. The impact of patients controlled analgesia undergoing orthopedic surgery. Braz J Anesthesiol. 2016;66:265-71.
-
76Konrath GA, Lock T, Goitz HT, Scheidler J. The use of cold therapy after anterior cruciate ligament reconstruction. A prospective, randomized study and literature review. Am J Sports Med. 1996 Sep-Oct;24(5):629-33.
-
77Koyonos L, Owsley K, Vollmer E, Limpisvasti O, Gambardella R. Preoperative cryotherapy use in anterior cruciate ligament reconstruction. J Knee Surg. 2014 Dec;27(6):479-84.
-
78Waterman B, Walker JJ, Swaims C, et al. The efficacy of combined cryotherapy and compression comparedwith cryotherapy alone following anterior cruciate ligament reconstruction. J Knee Surg. 2012 May;25(2):155-60.
-
79Kowal MA. Review of physiological effects of cryotherapy. J Orthop Sports Phys Ther. 1983;5(2):66-73.
-
80Drez D, Faust DC, Evans JP. Cryotherapy and nerve palsy. Am J Sports Med. 1981 Jul-Aug;9(4):256-7.
-
81McCarthy MR, Yates CK, Anderson MA, Yates-McCarthy JL. The effects of immediate continuous passive motion on pain during the inflammatory phase of soft tissue healing following anterior cruciate ligament reconstruction. J Orthop Sports Phys Ther. 1993;17(2):96-101.
-
82McCarthy M, Buxton B, Yates C. Effects of continuous passive motion on anterior laxity following ACL reconstruction with autogenous patellar tendon grafts. J Sport Rehabil. 1993;2:171-8.
-
83Smith T, Davies L. The efficacy of continuous passive motion after anterior cruciate ligament reconstruction: a systematic review. Phys Ther Sport. 2007;8:141-52.
-
84Richmond JC, Gladstone J, MacGillivray J. Continuous passive motion after arthroscopically assisted anterior cruciate ligament reconstruction: comparison of short- versus long-term use. Arthroscopy. 1991;7(1):39-44.
-
85Wright RW, Preston E, Fleming BC, et al. A systematic review of anterior cruciate ligament reconstruction rehabilitation: part I: continuous passive motion, early weight bearing, postoperative bracing, and home-based rehabilitation. J Knee Surg. 2008;21(3):217-24.
-
86Sluka K. The neurobiology of pain and foundations for electrical stimulation. En: Robinson A, Snyder-Mackler L (eds.). Clinical Electrophysiology. Baltimore, MD: Lippincott, Williams and Wilkins; 1995. p. 490.
-
87Jensen JE, Conn RR, Hazelrigg G, Hewett JE. The use of transcutaneous neural stimulation and isokinetic testing in arthroscopic knee surgery. Am J Sports Med. 1985;13(1):27-33.
Descargar artículo:
Licencia:
Este contenido es de acceso abierto (Open-Access) y se ha distribuido bajo los términos de la licencia Creative Commons CC BY-NC-ND (Reconocimiento-NoComercial-SinObraDerivada 4.0 Internacional) que permite usar, distribuir y reproducir en cualquier medio siempre que se citen a los autores y no se utilice para fines comerciales ni para hacer obras derivadas.
Comparte este contenido
En esta edición
- The importance of pain control
- Learning from pain to make the patient feel better
- Perioperative pain management in arthroscopic surgery of the shoulder
- Perioperative pain management in arthroscopy of the elbow
- Perioperative analgesia in arthroscopic surgery of the wrist and hand
- Perioperative analgesia in arthroscopy of the hip
- Perioperative analgesia in arthroscopic surgery of the knee
- Perioperative pain management in arthroscopy of the ankle
- Postoperative neuropathic pain in traumatology
- Capsaicin 179 mg patch application technique
- Type 3 SLAP: bucket handle tear
Más en PUBMED
Más en Google Scholar
Más en ORCID


Revista Española de Artroscopia y Cirugía Articular está distribuida bajo una licencia de Creative Commons Reconocimiento-NoComercial-SinObraDerivada 4.0 Internacional.